Three Tips To Help You Plan For CQC Infection Prevention and Control Visits
After an immensely challenging year for healthcare, the CQC continues to regulate care homes and hospitals by focussing primarily on infection prevention and control.
After each inspection, a report is made available on the CQC’s website – helping to reassure the public that services in their area have a reliable infection prevention and control strategy.
During an IPC inspection, the CQC will look at a number of key criteria to ensure that the required standards are being met. In particular, the inspectors will want to see that staff are properly trained to deal with outbreaks and that the correct procedures are in place.
So how can you ensure that you are managing infection control effectively in your facility, ahead of upcoming inspections?
The following tips will not only help you to be well prepared for your IPC inspection but will allow you to continue to prevent infection through winter and beyond.
CQC inspectors need to know that your staff training, practices and deployment will prevent transmission of infection.
Not sure if your infection control procedures are up to scratch?
1. Make sure your human waste disposal process is failsafe.
COVID-19 can be spread in all bodily secretions (except sweat), even if the individual is asymptomatic. This infection risk include includes stools and urine.
It is absolutely essential that the toileting needs of those in your care are managed impeccably – especially when bedpans, urinal bottles and commodes are in use.
Whether you choose to use disposable pulp products or a plastic re-usable equivalent, now is the time to ensure you’re making the best choice for cleaning or disposal – with clear procedures that your clinicians can follow with ease.
For those who use plastic or steel bedpans, the Panamatic Optima 2 washer disinfector is the ideal solution for high-demand wards or care settings.
Its features include:
- Large capacity, sanitising up to two bedpans and two urinals per cycle
- Antimicrobial surfaces to minimise cross infection and reduce the possibility of microbial growth
- Rapid eight-minute washing disinfection cycle which is eco-friendly and cost-effective
- Hands-free open and close technology using a foot cup or infrared sensor to minimise cross infection and provide ease of use
- Cold and hot wash cycles for an extra-thorough cleaning process before thermal disinfection.
For those who rely on a re-usable bedpan solution, the Panamatic Optima 2 will provide confidence that the risk of cross-infection between shared items is being managed responsibly.
But what about facilities that rely on single-use, disposable bedpans? What do they need to consider to satisfy CQC requirements?
For facilities such as these, a sluice room macerator is essential to dispose of waste – but not all macerators offer the same benefits. To truly protect against the threat of infection, you need a machine that will maximise uptime, minimise the risk of blockages, reduce contact points and be safe and simple for all of your clinicians to use.
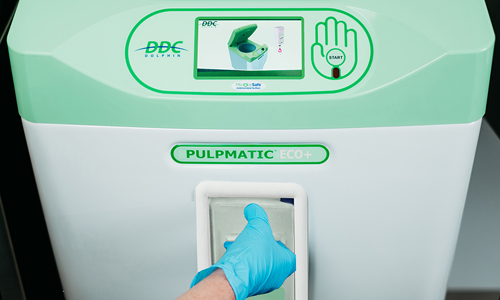
That’s why DDC Dolphin created the Pulpmatic Eco+.
This innovative machine incorporates more than 30 years' experience in sluice room product development, building on DDC Dolphin's expansive knowledge of user and facility requirements to launch a new standard in infection control.
Its features include:
- Cycle speed from 85 seconds with an improved drum design to save time
- Improved cutting performance with a new blade disc design which ensures pulp is macerated finely, quickly, and efficiently – reducing blockages
- Graphical user interface to provide animated operation instructions, cycle status information, as well as fault resolution guidance
- Click & Fit chemical system, an easy-load chemical cartridge system for improved antimicrobial kill rate efficacy
- MicrobeSafe+ antimicrobial technology in polymer surfaces – an antimicrobial and antifungal additive which stops the growth of harmful bacteria.
The Pulpmatic Eco+ is a powerful defence against the threat of infection. But as with all sluice room machinery, you must maintain it to ensure the best possible performance.
Don’t be caught out by unexpected machine breakdowns. Equip yourself with a care and maintenance package and ensure that you are always prepared for unexpected CQC visits.
Discover the benefits of world-class infection control, without obligation.
2. Ensure you have a strategy to keep your facility free from airborne bacteria and odour.
The Health and Social Care Act 2008 is enforced by CQC. Regulation 15 of the Act states:
“Premises and equipment should be visibly clean and free from odours that are offensive or unpleasant.”
To keep your facility smelling fresh and free from the threat of airborne bacteria, try adding a UV air purification system to your infection control armoury.
These powerful machines resolve persistent odour problems by destroying bacteria in the air and on hard surfaces. Not only do they destroy viruses such as influenza but E.coli, salmonella and similar bacteria as well.
A system such as the UVMATIC or UVMATIC Plus will equip you with benefits such as:
- 24/7 autonomous operation to provide total support to your existing cleaning regime
- Environmentally friendly protection, reducing the need for harsh chemical solutions to control bad odours and bacteria
- Minimal running costs – similar to a low-energy light bulb
- Minimal maintenance – allowing for cost-effective cleansing.
Not only will UV air purification keep your facility smelling clean and fresh on the day of your CQC inspection but it will provide a comfortable, healthy environment for your those in your care – all day, every day.
3. Equip yourself with a well-stocked store cupboard of infection-control consumables.
The right infection-control consumables should be powerful yet simple to use, allowing clinicians to maximise the effectiveness of cleaning procedures. As a result, they’ll be free to spend more time providing hands-on care.
The Hygenex range of consumable products does exactly that. It includes essential items such as:
- Maceratable & Flushable Wipes which are also biodegradable and compostable
- An Antimicrobial Coating System which is easy to apply and can be used to protect all your surfaces from major HCAIs including COVID-19
- Hand Sanitiser containing 76% Alcohol – it cleanses hands without the need for water.
DDC Dolphin can empower you to provide exceptional infection control and prepare for your next CQC inspection.
REGISTER FOR YOUR FREE INFECTION CONTROL AUDIT